Engineering a substitute provisional scaffold
After finding that premature provisional scaffold loss was a major mechanism behind the failure of the ACL to heal, we sought to develop a substitute provisional scaffold that is easily implanted and capable of providing the growth factors and enzymes required to optimize fibroblast and neurovascular ingrowth. Such a substitute scaffold would also need to withstand the physical conditions of the intra-articular environment.
Our experiments validated the hypothesis that multiple cytokines could stimulate cell proliferation, migration and collagen production in vitro; however, the improvements were modest.(1) This led us to look for a more physiologically relevant cytokine and growth factor delivery system. We focused on the use of platelets, since in classic studies of wound healing, platelet coagulation and platelet granule release are incipient events.
We designed and tested a strategy for delivering fresh platelets to an ACL wound using a stable carrier to see whether this would promote ACL healing that is similar to that of the MCL and other extraarticular ligaments.
We found that use of platelets, or platelet-rich plasma, was useful in stimulating key cellular behaviors in vitro. We determined that the concentration of growth factors released from platelet rich plasma was dependent on the platelet concentration(2)(Fig 1), and that the timing of that release was dependent on the activation method for the platelets(3;4;2). Thrombin resulted in an immediate release of the platelet-derived growth factors, while the use of collagen as an activator resulted in a more sustained release of multiple platelet derived growth factors(4) (Fig 2). We also found that keeping the platelets in their physiologic plasma resulted in an improved ability for the platelets to stimulate collagen synthesis by ACL fibroblasts(5) (Fig 3).
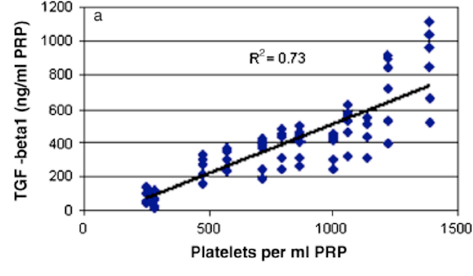 Figure 1: Platelet number versus growth factor release. When more platelets are incorporated into a blood clot or PRP clot, more growth factors are released from that clot. For the case of TGF-b, the relationship is linear as shown here. (Figure from Jacobson et al, Wound Repair and Regeneration, 2008, 16:370-378.)
Figure 1: Platelet number versus growth factor release. When more platelets are incorporated into a blood clot or PRP clot, more growth factors are released from that clot. For the case of TGF-b, the relationship is linear as shown here. (Figure from Jacobson et al, Wound Repair and Regeneration, 2008, 16:370-378.)
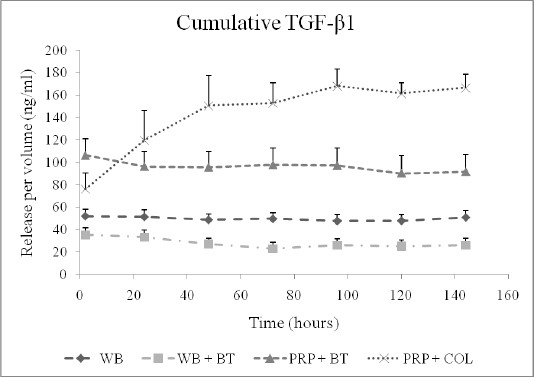 Figure 2: Cumulative TGF- β1 release over time measured in the supernatant surrounding four types of blood clots. The naturally clotting whole blood (WB), thrombin-activated whole blood (WB+BT) and thrombin-activated platelet-rich plasma (PRP+BT) clots released TGF- β1 at the two-hour time point and maintained the same level of cumulative TGF- β1 on the remaining six days. In contrast, the protein-activated platelet-rich plasma (PRP+COL) clots released a relatively small amount of TGF- β1 at the two-hour time point and showed an increase in the cumulative TGF- β1 levels in the supernatant between day one and five, after which the concentration remained constant. The shapes at each time point represent average values (n=9) of growth factor released and error bars are SEM. (Reprinted from Harrison et al, AJSM, 2011 39:729.)
Figure 2: Cumulative TGF- β1 release over time measured in the supernatant surrounding four types of blood clots. The naturally clotting whole blood (WB), thrombin-activated whole blood (WB+BT) and thrombin-activated platelet-rich plasma (PRP+BT) clots released TGF- β1 at the two-hour time point and maintained the same level of cumulative TGF- β1 on the remaining six days. In contrast, the protein-activated platelet-rich plasma (PRP+COL) clots released a relatively small amount of TGF- β1 at the two-hour time point and showed an increase in the cumulative TGF- β1 levels in the supernatant between day one and five, after which the concentration remained constant. The shapes at each time point represent average values (n=9) of growth factor released and error bars are SEM. (Reprinted from Harrison et al, AJSM, 2011 39:729.)
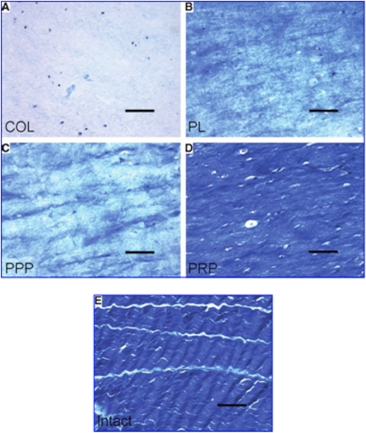 en in constructs of ACL cells and the addition of collagen only
en in constructs of ACL cells and the addition of collagen only
(A),platelets only (B),plasma without platelets (C), and platelets and plasma (D).
All are compared with the intact ACL (E) Scale bars: 100um. Note the similar intensity of staining in the intact and platelet+plasma groups. (Used with permission from Cheng et al, Tissue Engineering 16(5):1479-1489.)
References
- Meaney Murray, M, Rice, K, Wright, RJ, Spector, M. 2003. The effect of selected growth factors on human anterior cruciate ligament cell interactions with a three-dimensional collagen-GAG scaffold. J Orthop Res 21: 238-244.
- Jacobson, M, Fufa, D, Abreu, EL, et al. 2008. Platelets, but not erythrocytes, significantly affect cytokine release and scaffold contraction in a provisional scaffold model. Wound Repair Regen 16: 370-378.
- Fufa, D, Shealy, B, Jacobson, M, et al. 2008. Activation of platelet-rich plasma using soluble type I collagen. J Oral Maxillofac Surg 66: 684-690.
- Harrison, S, Vavken, P, Kevy, S, et al. 2011. Platelet Activation by Collagen Provides Sustained Release of Anabolic Cytokines. Am J Sports Med
- Cheng, M, Wang, H, Yoshida, R, Murray, MM. 2010. Platelets and Plasma Proteins Are Both Required to Stimulate Collagen Gene Expression by Anterior Cruciate Ligament Cells in Three-Dimensional Culture. Tissue Eng Part A.
